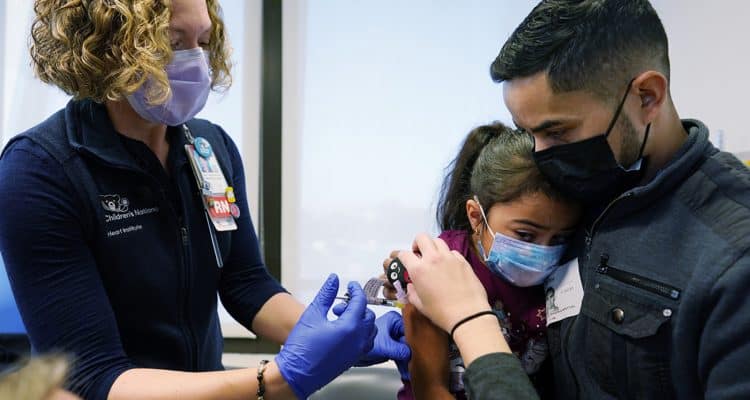
An FDA panel is recommending both Pfizer and Moderna’s COVID-19 vaccine for kids under 5. The recommendation applies to children as young as 6 months.
The 21-member panel’s decision is expected to be quickly authorized by the FDA. After that, a CDC panel will meet on Friday and Saturday. It’s expected they’ll also give the green light, sending the matter on to CDC Director Dr. Rochelle Walensky. Once she signs off, it’s possible that shots in arms can begin as soon as next Tuesday.
READ MORE: NY Mayor Eric Adams Doubles Back On Vaccine Rule For Pro Athletes, Performers
According to Cnet.com, the Moderna two-dose vaccine was submitted for approval for children 6 months through 5 years old, is 25 micrograms per dose, ¼ of each dose for adults.
While Pfizer’s vaccine is slightly different. It’s intended for kids 6 months through 4 years old and is administered as a series of three doses of 3 micrograms each, 1/10 of each dose for adults.